Search results
Search for "oxidation reactions" in Full Text gives 69 result(s) in Beilstein Journal of Organic Chemistry.
Enabling artificial photosynthesis systems with molecular recycling: A review of photo- and electrochemical methods for regenerating organic sacrificial electron donors
- Grace A. Lowe
Beilstein J. Org. Chem. 2023, 19, 1198–1215, doi:10.3762/bjoc.19.88
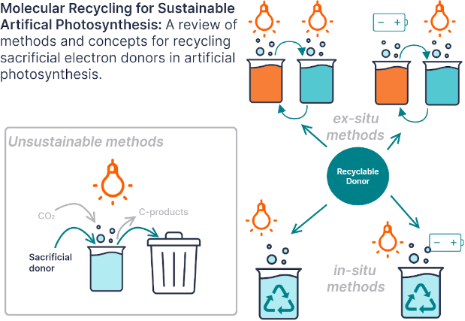
- material that shuttles electrons from one species to another through a series of chemically reversible reduction and oxidation reactions. In contrast, a sacrificial electron donor is a species that is oxidized to reduce another species and is consumed rather than regenerated. If a redox mediator is not re

Graphical Abstract

Figure 1: Diagram comparing the two reaction pathways for sacrificial electron donors (SD) in photocatalyzed ...

Figure 2: Diagram showing water-splitting systems developed by Girault, Scanlon, and co-workers that employ i...

Figure 3: Diagram illustrating the transfer of electrons in a photocatalytic particulate suspensions Z-scheme...

Figure 4: A. Structures of the molecules represented in part B. The numbers in brackets correspond to the com...

Figure 5: A. Structures of the molecules represented in part B. The numbers in brackets correspond to the com...
Selective and scalable oxygenation of heteroatoms using the elements of nature: air, water, and light
- Damiano Diprima,
- Hannes Gemoets,
- Stefano Bonciolini and
- Koen Van Aken
Beilstein J. Org. Chem. 2023, 19, 1146–1154, doi:10.3762/bjoc.19.82
- flow using the HANU flow reactor, indicating scalability and improving safety. Keywords: catalyst-free; flow chemistry; oxygen; photochemistry; sustainable oxidation; Introduction Oxidation reactions are widely used in the chemical industry, but are often problematic due to challenges with

Graphical Abstract

Scheme 1: Oxidation of heteroatoms.

Scheme 2: Graphical representation comparing A electrochemistry and B photoredox catalysis using a semiconduc...

Figure 1: Study of additives. A) Effect of the addition of 1 equiv of various acids and bases to the standard...

Scheme 3: Substrate scope with reaction times and isolated yields. 1 mmol (1 equiv) substrate was reacted in ...

Scheme 4: Setup used in the flow experiment for the triphenylphosphine oxidation.

Scheme 5: Proposed extra alternative pathway.
Redox-active molecules as organocatalysts for selective oxidative transformations – an unperceived organocatalysis field
- Elena R. Lopat’eva,
- Igor B. Krylov,
- Dmitry A. Lapshin and
- Alexander O. Terent’ev
Beilstein J. Org. Chem. 2022, 18, 1672–1695, doi:10.3762/bjoc.18.179
- substrates. DDQ is a popular mediator for oxidation reactions. It has been used for intramolecular dehydrogenative C–C bond formation between aromatic groups [127]. Using this method, the formation of polyaromatic systems was achieved in good yields (Scheme 26). The cross-dehydrogenative C–N coupling of

Graphical Abstract

Scheme 1: Organocatalysis classification used in the present perspective.

Scheme 2: Oxidative processes catalyzed by amines.

Scheme 3: N-Heterocyclic carbene (NHC) catalysis in oxidative functionalization of aldehydes.

Scheme 4: Examples of asymmetric oxidative processes catalyzed by chiral Brønsted acids.

Scheme 5: Asymmetric aerobic α-hydroxylation of lactams under phase-transfer organocatalysis conditions emplo...

Scheme 6: Selective CH-oxidation of methylarenes to aldehydes or carboxylic acids.

Scheme 7: An example of the regioselective CH-amination by a sterically hindered imide-N-oxyl radical precurs...

Scheme 8: CH-amination of ethylbenzene and CH-fluorination of aldehydes catalyzed by N-hydroxybenzimidazoles,...

Scheme 9: Mixed hetero-/homogeneous TiO2/N-hydroxyimide photocatalysis in the selective benzylic oxidation.

Scheme 10: Electrochemical benzylic iodination and benzylation of pyridine by benzyl iodides generated in situ...

Scheme 11: Electrochemical oxidative C–O/C–N coupling of alkylarenes with NHPI. Electrolysis conditions: Const...

Scheme 12: Chemoselective alcohol oxidation catalyzed by TEMPO.

Scheme 13: ABNO-catalyzed oxidative C–N coupling of primary alcohols with primary amines.

Scheme 14: ACT-catalyzed electrochemical oxidation of primary alcohols and aldehydes to carboxylic acids.

Scheme 15: Electrocatalytic oxidation of benzylic alcohols by a TEMPO derivative immobilized on a graphite ano...

Scheme 16: Electrochemical oxidation of carbamates of cyclic amines to lactams and oxidative cyanation of amin...

Scheme 17: Hydrogen atom transfer (HAT) and single-electron transfer (SET) as basic principles of amine cation...

Scheme 18: Electrochemical quinuclidine-catalyzed oxidation involving unactivated C–H bonds.

Scheme 19: DABCO-mediated photocatalytic C–C cross-coupling involving aldehyde C–H bond cleavage.

Scheme 20: DABCO-derived cationic catalysts in inactivated C–H bond cleavage for alkyl radical addition to ele...

Scheme 21: Electrochemical diamination and dioxygenation of vinylarenes catalyzed by triarylamines.

Scheme 22: Electrochemical benzylic oxidation mediated by triarylimidazoles.

Scheme 23: Thiyl radical-catalyzed CH-arylation of allylic substrates by aryl cyanides.

Scheme 24: Synthesis of redox-active alkyl tetrafluoropyridinyl sulfides by unactivated C–H bond cleavage by t...

Scheme 25: Main intermediates in quinone oxidative organocatalysis.

Scheme 26: Electrochemical DDQ-catalyzed intramolecular dehydrogenative aryl–aryl coupling.

Scheme 27: DDQ-mediated cross-dehydrogenative C–N coupling of benzylic substrates with azoles.

Scheme 28: Biomimetic o-quinone-catalyzed benzylic alcohol oxidation.

Scheme 29: Electrochemical synthesis of secondary amines by oxidative coupling of primary amines and benzylic ...

Scheme 30: General scheme of dioxirane and oxaziridine oxidative organocatalysis.

Scheme 31: Dioxirane organocatalyzed CH-hydroxylation involving aliphatic C(sp3)–H bonds.

Scheme 32: Enantioselective hydroxylation of CH-acids catalyzed by chiral oxaziridines.

Scheme 33: Iodoarene-organocatalyzed vinylarene diamination.

Scheme 34: Iodoarene-organocatalyzed asymmetric CH-hydroxylation of benzylic substrates.

Scheme 35: Iodoarene-organocatalyzed asymmetric difluorination of alkenes with migration of aryl or methyl gro...

Scheme 36: Examples of 1,2-diiodo-4,5-dimethoxybenzene-catalyzed electrochemical oxidative heterocyclizations.

Scheme 37: Electrochemical N-ammonium ylide-catalyzed CH-oxidation.

Scheme 38: Oxidative dimerization of aryl- and alkenylmagnesium compounds catalyzed by quinonediimines.

Scheme 39: FLP-catalyzed dehydrogenation of N-substituted indolines.
Electro-conversion of cumene into acetophenone using boron-doped diamond electrodes
- Mana Kitano,
- Tsuyoshi Saitoh,
- Shigeru Nishiyama,
- Yasuaki Einaga and
- Takashi Yamamoto
Beilstein J. Org. Chem. 2022, 18, 1154–1158, doi:10.3762/bjoc.18.119
- a sustainable, scalable, and cost-efficient protocol; a specific catalyst is not required, and reagent waste can be avoided. In addition, the present work offers new perspectives for an electrosynthetic strategy toward oxidation reactions of aromatic alkyls. Experimental General protocol for electro

Graphical Abstract

Figure 1: (a) Cyclic voltammograms of a BDD electrode in MeCN solution containing cumene (1; 5 mM) and Et4NClO...

Figure 2: Proposed reaction mechanism of electro-conversion of cumene (1) into acetophenone (3).
Substituent effect on TADF properties of 2-modified 4,6-bis(3,6-di-tert-butyl-9-carbazolyl)-5-methylpyrimidines
- Irina Fiodorova,
- Tomas Serevičius,
- Rokas Skaisgiris,
- Saulius Juršėnas and
- Sigitas Tumkevicius
Beilstein J. Org. Chem. 2022, 18, 497–507, doi:10.3762/bjoc.18.52
- pyrimidine–carbazole TADF emitters bearing different substituents in position 2 of the pyrimidine moiety were successfully prepared by using Liebeskind–Srogl cross-coupling, hydrogenolysis, oxidation reactions of 4,6-bis(3,6-di-tert-butyl-9-carbazolyl)-5-methyl-2-methylthiopyrimidine and following

Graphical Abstract

Figure 1: 2-Modified 4,6-bis(3,6-di-tert-butyl-9H-carbazol-9-yl)-5-methylpyrimidines.

Scheme 1: Synthesis of 4,6-bis(3,6-di-tert-butyl-9H-carbazol-9-yl)-5-methyl-2-substituted pyrimidines 1–6. Re...

Figure 2: HOMO and LUMO spatial distributions of carbazole–pyrimidine TADF compounds.

Figure 3: Absorption (grey lines), fluorescence (black lines) and 10K phosphorescence (red lines) spectra of ...

Figure 4: Fluorescence decay transients of 1 wt % PMMA films of carbazole–pyrimidine TADF compounds in oxygen...
Menadione: a platform and a target to valuable compounds synthesis
- Acácio S. de Souza,
- Ruan Carlos B. Ribeiro,
- Dora C. S. Costa,
- Fernanda P. Pauli,
- David R. Pinho,
- Matheus G. de Moraes,
- Fernando de C. da Silva,
- Luana da S. M. Forezi and
- Vitor F. Ferreira
Beilstein J. Org. Chem. 2022, 18, 381–419, doi:10.3762/bjoc.18.43
- epoxides are formed through oxidation reactions in vivo, that occur in protein processes dependent on vitamin K [96][97]. Dwyer and co-workers described a procedure using sugar-derived hydroperoxides for the synthesis of epoxides in the presence of 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) as base [98][99

Graphical Abstract

Figure 1: Natural bioactive naphthoquinones.

Figure 2: Chemical structures of vitamins K.

Figure 3: Redox cycle of menadione.

Scheme 1: Selected approaches for menadione synthesis using silver(I) as a catalyst.

Scheme 2: Methylation approaches for the preparation of menadione from 1,4-naphthoquinone using tert-butyl hy...

Scheme 3: Methylation approach of 1,4-naphthoquinone using i) rhodium complexes/methylboronic acid and ii) bi...

Scheme 4: Synthesis of menadione (10) from itaconic acid.

Scheme 5: Menadione synthesis via Diels–Alder reaction.

Scheme 6: Synthesis of menadione (10) using p-cresol as a synthetic precursor.

Scheme 7: Synthesis of menadione (10) via demethoxycarbonylating annulation of methyl methacrylate.

Scheme 8: Furan 34 used as a diene in a Diels–Alder reaction for the synthesis of menadione (10).

Scheme 9: o-Toluidine as a dienophile in a Diels–Alder reaction for the synthesis of menadione (10).

Scheme 10: Representation of electrochemical synthesis of menadione.

Figure 4: Reaction sites and reaction types of menadione as substrate.

Scheme 11: DBU-catalyzed epoxidation of menadione (10).

Scheme 12: Phase-transfer catalysis for the epoxidation of menadione.

Scheme 13: Menadione epoxidation using a hydroperoxide derived from (+)-norcamphor.

Scheme 14: Enantioselective Diels–Alder reaction for the synthesis of asymmetric quinone 50 catalyzed by a chi...

Scheme 15: Optimized reaction conditions for the synthesis of anthra[9,1-bc]pyranone.

Scheme 16: Synthesis of anthra[9,1-bc]furanone, anthra[9,1-bc]pyridine, and anthra[9,1-bc]pyrrole derivatives.

Scheme 17: Synthesis of derivatives employing protected trienes.

Scheme 18: Synthesis of cyclobutene derivatives of menadione.

Scheme 19: Menadione reduction reactions using sodium hydrosulfite.

Scheme 20: Green methodology for menadiol synthesis and pegylation.

Scheme 21: Menadione reduction by 5,6-O-isopropylidene-ʟ-ascorbic acid under UV light irradiation.

Scheme 22: Selected approaches of menadione hydroacetylation to diacetylated menadiol.

Scheme 23: Thiele–Winter reaction catalyzed by Bi(OTf)3.

Scheme 24: Carbonyl condensation of menadione using resorcinol and a hydrazone derivative.

Scheme 25: Condensation reaction of menadione with thiosemicarbazide.

Scheme 26: Condensation reaction of menadione with acylhydrazides.

Scheme 27: Menadione derivatives functionalized with organochalcogens.

Scheme 28: Synthesis of selenium-menadione conjugates derived from chloromethylated menadione 84.

Scheme 29: Menadione alkylation by the Kochi–Anderson method.

Scheme 30: Menadione alkylation by diacids.

Scheme 31: Menadione alkylation by heterocycles-substituted carboxylic acids.

Scheme 32: Menadione alkylation by bromoalkyl-substituted carboxylic acids.

Scheme 33: Menadione alkylation by complex carboxylic acids.

Scheme 34: Kochi–Anderson method variations for the menadione alkylation via oxidative decarboxylation of carb...

Scheme 35: Copper-catalyzed menadione alkylation via free radicals.

Scheme 36: Nickel-catalyzed menadione cyanoalkylation.

Scheme 37: Iron-catalyzed alkylation of menadione.

Scheme 38: Selected approaches to menadione alkylation.

Scheme 39: Menadione acylation by photo-Friedel–Crafts acylation reported by Waske and co-workers.

Scheme 40: Menadione acylation by Westwood procedure.

Scheme 41: Synthesis of 3-benzoylmenadione via metal-free TBAI/TBHP system.

Scheme 42: Michael-type addition of amines to menadione reported by Kallmayer.

Scheme 43: Synthesis of amino-menadione derivatives using polyalkylamines.

Scheme 44: Selected examples for the synthesis of different amino-substituted menadione derivatives.

Scheme 45: Selected examples of Michael-type addition of complex amines to menadione (10).

Scheme 46: Addition of different natural α-amino acids to menadione.

Scheme 47: Michael-type addition of amines to menadione using silica-supported perchloric acid.

Scheme 48: Indolylnaphthoquinone or indolylnaphthalene-1,4-diol synthesis reported by Yadav et al.

Scheme 49: Indolylnaphthoquinone synthesis reported by Tanoue and co-workers.

Scheme 50: Indolylnaphthoquinone synthesis from menadione by Escobeto-González and co-workers.

Scheme 51: Synthesis of menadione analogues functionalized with thiols.

Scheme 52: Synthesis of menadione-derived symmetrical derivatives through reaction with dithiols.

Scheme 53: Mercaptoalkyl acids as nucleophiles in Michael-type addition reaction to menadione.

Scheme 54: Reactions of menadione (10) with cysteine derivatives for the synthesis of quinoproteins.

Scheme 55: Synthesis of menadione-glutathione conjugate 152 by Michael-type addition.
On the application of 3d metals for C–H activation toward bioactive compounds: The key step for the synthesis of silver bullets
- Renato L. Carvalho,
- Amanda S. de Miranda,
- Mateus P. Nunes,
- Roberto S. Gomes,
- Guilherme A. M. Jardim and
- Eufrânio N. da Silva Júnior
Beilstein J. Org. Chem. 2021, 17, 1849–1938, doi:10.3762/bjoc.17.126

- have been used as additives in these reactions and are suggested to act as ligands, assist proton transfer and promote the formation of oligovanadates by decreasing the pH value of the solution. The mechanisms of some vanadium-mediated oxidation reactions of alkanes have been studied, most of them

Graphical Abstract

Scheme 1: Schematic overview of transition metals studied in C–H activation processes.

Scheme 2: (A) Known biological activities related to benzimidazole-based compounds; (B and C) an example of a...

Scheme 3: (A) Known biological activities related to quinoline-based compounds; (B and C) an example of a sca...

Scheme 4: (A) Known biological activities related to sulfur-containing compounds; (B and C) an example of a s...

Scheme 5: (A) Known biological activities related to aminoindane derivatives; (B and C) an example of a scand...

Scheme 6: (A) Known biological activities related to norbornane derivatives; (B and C) an example of a scandi...

Scheme 7: (A) Known biological activities related to aniline derivatives; (B and C) an example of a titanium-...

Scheme 8: (A) Known biological activities related to cyclohexylamine derivatives; (B) an example of an intram...

Scheme 9: (A) Known biologically active benzophenone derivatives; (B and C) photocatalytic oxidation of benzy...

Scheme 10: (A) Known bioactive fluorine-containing compounds; (B and C) vanadium-mediated C(sp3)–H fluorinatio...

Scheme 11: (A) Known biologically active Lythraceae alkaloids; (B) synthesis of (±)-decinine (30).

Scheme 12: (A) Synthesis of (R)- and (S)-boehmeriasin (31); (B) synthesis of phenanthroindolizidines by vanadi...

Scheme 13: (A) Known bioactive BINOL derivatives; (B and C) vanadium-mediated oxidative coupling of 2-naphthol...

Scheme 14: (A) Known antiplasmodial imidazopyridazines; (B) practical synthesis of 41.

Scheme 15: (A) Gold-catalyzed drug-release mechanism using 2-alkynylbenzamides; (B and C) chromium-mediated al...

Scheme 16: (A) Examples of anti-inflammatory benzaldehyde derivatives; (B and C) chromium-mediated difunctiona...

Scheme 17: (A and B) Manganese-catalyzed chemoselective intramolecular C(sp3)–H amination; (C) late-stage modi...

Scheme 18: (A and B) Manganese-catalyzed C(sp3)–H amination; (C) late-stage modification of a leelamine deriva...

Scheme 19: (A) Known bioactive compounds containing substituted N-heterocycles; (B and C) manganese-catalyzed ...

Scheme 20: (A) Known indoles that present GPR40 full agonist activity; (B and C) manganese-catalyzed C–H alkyl...

Scheme 21: (A) Examples of known biaryl-containing drugs; (B and C) manganese-catalyzed C–H arylation through ...

Scheme 22: (A) Known zidovudine derivatives with potent anti-HIV properties; (B and C) manganese-catalyzed C–H...

Scheme 23: (A and B) Manganese-catalyzed C–H organic photo-electrosynthesis; (C) late-stage modification.

Scheme 24: (A) Example of a known antibacterial silylated dendrimer; (B and C) manganese-catalyzed C–H silylat...

Scheme 25: (A and B) Fe-based small molecule catalyst applied for selective aliphatic C–H oxidations; (C) late...

Scheme 26: (A) Examples of naturally occurring gracilioethers; (B) the first total synthesis of gracilioether ...

Scheme 27: (A and B) Selective aliphatic C–H oxidation of amino acids; (C) late-stage modification of proline-...

Scheme 28: (A) Examples of Illicium sesquiterpenes; (B) first chemical synthesis of (+)-pseudoanisatin (80) in...

Scheme 29: (A and B) Fe-catalyzed deuteration; (C) late-stage modification of pharmaceuticals.

Scheme 30: (A and B) Biomimetic Fe-catalyzed aerobic oxidation of methylarenes to benzaldehydes (PMHS, polymet...

Scheme 31: (A) Known tetrahydroquinolines with potential biological activities; (B and C) redox-selective Fe c...

Scheme 32: (A) Known drugs containing a benzofuran unit; (B and C) Fe/Cu-catalyzed tandem O-arylation to acces...

Scheme 33: (A) Known azaindolines that act as M4 muscarinic acetylcholine receptor agonists; (B and C) intramo...

Scheme 34: (A) Known indolinones with anticholinesterase activity; (B and C) oxidative C(sp3)–H cross coupling...

Scheme 35: (A and B) Cobalt-catalyzed C–H alkenylation of C-3-peptide-containing indoles; (C) derivatization b...

Scheme 36: (A) Cobalt-Cp*-catalyzed C–H methylation of known drugs; (B and C) scope of the o-methylated deriva...

Scheme 37: (A) Known lasalocid A analogues; (B and C) three-component cobalt-catalyzed C–H bond addition; (D) ...

Scheme 38: (A and B) Cobalt-catalyzed C(sp2)–H amidation of thiostrepton.

Scheme 39: (A) Known 4H-benzo[d][1,3]oxazin-4-one derivatives with hypolipidemic activity; (B and C) cobalt-ca...

Scheme 40: (A and B) Cobalt-catalyzed C–H arylation of pyrrole derivatives; (C) application for the synthesis ...

Scheme 41: (A) Known 2-phenoxypyridine derivatives with potent herbicidal activity; (B and C) cobalt-catalyzed...

Scheme 42: (A) Natural cinnamic acid derivatives; (B and C) cobalt-catalyzed C–H carboxylation of terminal alk...

Scheme 43: (A and B) Cobalt-catalyzed C–H borylation; (C) application to the synthesis of flurbiprofen.

Scheme 44: (A) Benzothiazoles known to present anticonvulsant activities; (B and C) cobalt/ruthenium-catalyzed...

Scheme 45: (A and B) Cobalt-catalyzed oxygenation of methylene groups towards ketone synthesis; (C) synthesis ...

Scheme 46: (A) Known anticancer tetralone derivatives; (B and C) cobalt-catalyzed C–H difluoroalkylation of ar...

Scheme 47: (A and B) Cobalt-catalyzed C–H thiolation; (C) application in the synthesis of quetiapine (153).

Scheme 48: (A) Known benzoxazole derivatives with anticancer, antifungal, and antibacterial activities; (B and...

Scheme 49: (A and B) Cobalt-catalyzed C–H carbonylation of naphthylamides; (C) BET inhibitors 158 and 159 tota...

Scheme 50: (A) Known bioactive pyrrolo[1,2-a]quinoxalin-4(5H)-one derivatives; (B and C) cobalt-catalyzed C–H ...

Scheme 51: (A) Known antibacterial cyclic sulfonamides; (B and C) cobalt-catalyzed C–H amination of propargyli...

Scheme 52: (A and B) Cobalt-catalyzed intramolecular 1,5-C(sp3)–H amination; (C) late-stage functionalization ...

Scheme 53: (A and B) Cobalt-catalyzed C–H/C–H cross-coupling between benzamides and oximes; (C) late-state syn...

Scheme 54: (A) Known anticancer natural isoquinoline derivatives; (B and C) cobalt-catalyzed C(sp2)–H annulati...

Scheme 55: (A) Enantioselective intramolecular nickel-catalyzed C–H activation; (B) bioactive obtained motifs;...

Scheme 56: (A and B) Nickel-catalyzed α-C(sp3)–H arylation of ketones; (C) application of the method using kno...

Scheme 57: (A and B) Nickel-catalyzed C(sp3)–H acylation of pyrrolidine derivatives; (C) exploring the use of ...

Scheme 58: (A) Nickel-catalyzed C(sp3)–H arylation of dioxolane; (B) library of products obtained from biologi...

Scheme 59: (A) Intramolecular enantioselective nickel-catalyzed C–H cycloalkylation; (B) product examples, inc...

Scheme 60: (A and B) Nickel-catalyzed C–H deoxy-arylation of azole derivatives; (C) late-stage functionalizati...

Scheme 61: (A and B) Nickel-catalyzed decarbonylative C–H arylation of azole derivatives; (C) application of t...

Scheme 62: (A and B) Another important example of nickel-catalyzed C–H arylation of azole derivatives; (C) app...

Scheme 63: (A and B) Another notable example of a nickel-catalyzed C–H arylation of azole derivatives; (C) lat...

Scheme 64: (A and B) Nickel-based metalorganic framework (MOF-74-Ni)-catalyzed C–H arylation of azole derivati...

Scheme 65: (A) Known commercially available benzothiophene-based drugs; (B and C) nickel-catalyzed C–H arylati...

Scheme 66: (A) Known natural tetrahydrofuran-containing substances; (B and C) nickel-catalyzed photoredox C(sp3...

Scheme 67: (A and B) Another notable example of a nickel-catalyzed photoredox C(sp3)–H alkylation/arylation; (...

Scheme 68: (A) Electrochemical/nickel-catalyzed C–H alkoxylation; (B) achieved scope, including three using na...

Scheme 69: (A) Enantioselective photoredox/nickel catalyzed C(sp3)–H arylation; (B) achieved scope, including ...

Scheme 70: (A) Known commercially available trifluoromethylated drugs; (B and C) nickel-catalyzed C–H trifluor...

Scheme 71: (A and B) Stereoselective nickel-catalyzed C–H difluoroalkylation; (C) late-stage functionalization...

Scheme 72: (A) Cu-mediated ortho-amination of oxalamides; (B) achieved scope, including derivatives obtained f...

Scheme 73: (A) Electro-oxidative copper-mediated amination of 8-aminoquinoline-derived amides; (B) achieved sc...

Scheme 74: (A and B) Cu(I)-mediated C–H amination with oximes; (C) derivatization using telmisartan (241) as s...

Scheme 75: (A and B) Cu-mediated amination of aryl amides using ammonia; (C) late-stage modification of proben...

Scheme 76: (A and B) Synthesis of purine nucleoside analogues using copper-mediated C(sp2)–H activation.

Scheme 77: (A) Copper-mediated annulation of acrylamide; (B) achieved scope, including the synthesis of the co...

Scheme 78: (A) Known bioactive compounds containing a naphthyl aryl ether motif; (B and C) copper-mediated eth...

Scheme 79: (A and B) Cu-mediated alkylation of N-oxide-heteroarenes; (C) late-stage modification.

Scheme 80: (A) Cu-mediated cross-dehydrogenative coupling of polyfluoroarenes and alkanes; (B) scope from know...

Scheme 81: (A) Known anticancer acrylonitrile compounds; (B and C) Copper-mediated cyanation of unactivated al...

Scheme 82: (A) Cu-mediated radiofluorination of 8-aminoquinoline-derived aryl amides; (B) achieved scope, incl...

Scheme 83: (A) Examples of natural β-carbolines; (B and C) an example of a zinc-catalyzed C–H functionalizatio...

Scheme 84: (A) Examples of anticancer α-aminophosphonic acid derivatives; (B and C) an example of a zinc-catal...
Cerium-photocatalyzed aerobic oxidation of benzylic alcohols to aldehydes and ketones
- Girish Suresh Yedase,
- Sumit Kumar,
- Jessica Stahl,
- Burkhard König and
- Veera Reddy Yatham
Beilstein J. Org. Chem. 2021, 17, 1727–1732, doi:10.3762/bjoc.17.121
- acting as the terminal oxidant [13][14][15][16][17][18][19][20][21][22][23][24][25][26][27][28][29][30][31][32][33]. Most aerobic oxidation reactions utilize either metal complexes and nanoparticles or persistent radical reagents as catalysts [21]. In the past ten years, visible light-induced

Graphical Abstract

Scheme 1: Photocatalyzed aerobic oxidation of aromatic alcohols.

Scheme 2: Substrate scope. Reaction conditions as given in Table 1 (entry 1). Yields are isolated yields, average of...

Scheme 3: Selective oxidation of 3-bromobenzyl alcohol in the presence of 3-phenylpropanol. Compound 1af was ...

Figure 1: Mechanistic studies. (A): UV–vis spectra of the CeIV(OBn)Cln complex in CH3CN under blue light irra...
A comprehensive review of flow chemistry techniques tailored to the flavours and fragrances industries
- Guido Gambacorta,
- James S. Sharley and
- Ian R. Baxendale
Beilstein J. Org. Chem. 2021, 17, 1181–1312, doi:10.3762/bjoc.17.90

Graphical Abstract

Figure 1: Representative shares of the global F&F market (2018) segmented on their applications [1].

Figure 2: General structure of an international fragrance company [2].

Figure 3: The Michael Edwards fragrance wheel.

Figure 4: Examples of oriental (1–3), woody (4–7), fresh (8–10), and floral (11 and 12) notes.

Figure 5: A basic depiction of batch vs flow.

Scheme 1: Examples of reactions for which flow processing outperforms batch.

Scheme 2: Some industrially important aldol-based transformations.

Scheme 3: Biphasic continuous aldol reactions of acetone and various aldehydes.

Scheme 4: Aldol synthesis of 43 in flow using LiHMDS as the base.

Scheme 5: A semi-continuous synthesis of doravirine (49) involving a key aldol reaction.

Scheme 6: Enantioselective aldol reaction using 5-(pyrrolidin-2-yl)tetrazole (51) as catalyst in a microreact...

Scheme 7: Gröger's example of asymmetric aldol reaction in aqueous media.

Figure 6: Immobilised reagent column reactor types.

Scheme 8: Photoinduced thiol–ene coupling preparation of silica-supported 5-(pyrrolidin-2-yl)tetrazole 63 and...

Scheme 9: Continuous-flow approach for enantioselective aldol reactions using the supported catalyst 67.

Scheme 10: Ötvös’ employment of a solid-supported peptide aldol catalyst in flow.

Scheme 11: The use of proline tetrazole packed in a column for aldol reaction between cyclohexanone (65) and 2...

Scheme 12: Schematic diagram of an aminosilane-grafted Si-Zr-Ti/PAI-HF reactor for continuous-flow aldol and n...

Scheme 13: Continuous-flow condensation for the synthesis of the intermediate 76 to nabumetone (77) and Microi...

Scheme 14: Synthesis of ψ-Ionone (80) in continuous-flow via aldol condensation between citral (79) and aceton...

Scheme 15: Synthesis of β-methyl-ionones (83) from citral (79) in flow. The steps are separately described, an...

Scheme 16: Continuous-flow synthesis of 85 from 84 described by Gavriilidis et al.

Scheme 17: Continuous-flow scCO2 apparatus for the synthesis of 2-methylpentanal (87) and the self-condensed u...

Scheme 18: Chen’s two-step flow synthesis of coumarin (90).

Scheme 19: Pechmann condensation for the synthesis of 7-hydroxyxcoumarin (93) in flow. The setup extended to c...

Scheme 20: Synthesis of the dihydrojasmonate 35 exploiting nitro derivative proposed by Ballini et al.

Scheme 21: Silica-supported amines as heterogeneous catalyst for nitroaldol condensation in flow.

Scheme 22: Flow apparatus for the nitroaldol condensation of p-hydroxybenzaldehyde (102) to nitrostyrene 103 a...

Scheme 23: Nitroaldol reaction of 64 to 105 employing a quaternary ammonium functionalised PANF.

Scheme 24: Enantioselective nitroaldol condensation for the synthesis of 108 under flow conditions.

Scheme 25: Enatioselective synthesis of 1,2-aminoalcohol 110 via a copper-catalysed nitroaldol condensation.

Scheme 26: Examples of Knoevenagel condensations applied for fragrance components.

Scheme 27: Flow apparatus for Knoevenagel condensation described in 1989 by Venturello et al.

Scheme 28: Knoevenagel reaction using a coated multichannel membrane microreactor.

Scheme 29: Continuous-flow apparatus for Knoevenagel condensation employing sugar cane bagasse as support deve...

Scheme 30: Knoevenagel reaction for the synthesis of 131–135 in flow using an amine-functionalised silica gel. ...

Scheme 31: Continuous-flow synthesis of compound 137, a key intermediate for the synthesis of pregabalin (138)...

Scheme 32: Continuous solvent-free apparatus applied for the synthesis of compounds 140–143 using a TSE. Throu...

Scheme 33: Lewis et al. developed a spinning disc reactor for Darzens condensation of 144 and a ketone to furn...

Scheme 34: Some key industrial applications of conjugate additions in the F&F industry.

Scheme 35: Continuous-flow synthesis of 4-(2-hydroxyethyl)thiomorpholine 1,1-dioxide (156) via double conjugat...

Scheme 36: Continuous-flow system for Michael addition using CsF on alumina as the catalyst.

Scheme 37: Calcium chloride-catalysed asymmetric Michael addition using an immobilised chiral ligand.

Scheme 38: Continuous multistep synthesis for the preparation of (R)-rolipram (173). Si-NH2: primary amine-fun...

Scheme 39: Continuous-flow Michael addition using ion exchange resin Amberlyst® A26.

Scheme 40: Preparation of the heterogeneous catalyst 181 developed by Paixão et al. exploiting Ugi multicompon...

Scheme 41: Continuous-flow system developed by the Paixão’s group for the preparation of Michael asymmetric ad...

Scheme 42: Continuous-flow synthesis of nitroaldols catalysed by supported catalyst 184 developed by Wennemers...

Scheme 43: Heterogenous polystyrene-supported catalysts developed by Pericàs and co-workers.

Scheme 44: PANF-supported pyrrolidine catalyst for the conjugate addition of cyclohexanone (65) and trans-β-ni...

Scheme 45: Synthesis of (−)-paroxetine precursor 195 developed by Ötvös, Pericàs, and Kappe.

Scheme 46: Continuous-flow approach for the 5-step synthesis of (−)-oseltamivir (201) as devised by Hayashi an...

Scheme 47: Continuous-flow enzyme-catalysed Michael addition.

Scheme 48: Continuous-flow copper-catalysed 1,4 conjugate addition of Grignard reagents to enones. Reprinted w...

Scheme 49: A collection of commonly encountered hydrogenation reactions.

Figure 7: The ThalesNano H-Cube® continuous-flow hydrogenator.

Scheme 50: Chemoselective reduction of an α,β-unsaturated ketone using the H-Cube® reactor.

Scheme 51: Incorporation of Lindlar’s catalyst into the H-Cube® reactor for the reduction of an alkyne.

Scheme 52: Continuous-flow semi-hydrogenation of alkyne 208 to 209 using SACs with H-Cube® system.

Figure 8: The standard setups for tube-in-tube gas–liquid reactor units.

Scheme 53: Homogeneous hydrogenation of olefins using a tube-in-tube reactor setup.

Scheme 54: Recyclable heterogeneous flow hydrogenation system.

Scheme 55: Leadbeater’s reverse tube-in-tube hydrogenation system for olefin reductions.

Scheme 56: a) Hydrogenation using a Pd-immobilised microchannel reactor (MCR) and b) a representation of the i...

Scheme 57: Hydrogenation of alkyne 238 exploiting segmented flow in a Pd-immobilised capillary reactor.

Scheme 58: Continuous hydrogenation system for the preparation of cyrene (241) from (−)-levoglucosenone (240).

Scheme 59: Continuous hydrogenation system based on CSMs developed by Hornung et al.

Scheme 60: Chemoselective reduction of carbonyls (ketones over aldehydes) in flow.

Scheme 61: Continuous system for the semi-hydrogenation of 256 and 258, developed by Galarneau et al.

Scheme 62: Continuous synthesis of biodiesel fuel 261 from lignin-derived furfural acetone (260).

Scheme 63: Continuous synthesis of γ-valerolacetone (263) via CTH developed by Pineda et al.

Scheme 64: Continuous hydrogenation of lignin-derived biomass (products 265, 266, and 267) using a sustainable...

Scheme 65: Ru/C or Rh/C-catalysed hydrogenation of arene in flow as developed by Sajiki et al.

Scheme 66: Polysilane-immobilized Rh–Pt-catalysed hydrogenation of arenes in flow by Kobayashi et al.

Scheme 67: High-pressure in-line mixing of H2 for the asymmetric reduction of 278 at pilot scale with a 73 L p...

Figure 9: Picture of the PFR employed at Eli Lilly & Co. for the continuous hydrogenation of 278 [287]. Reprinted ...

Scheme 68: Continuous-flow asymmetric hydrogenation using Oppolzer's sultam 280 as chiral auxiliary.

Scheme 69: Some examples of industrially important oxidation reactions in the F&F industry. CFL: compact fluor...

Scheme 70: Gold-catalysed heterogeneous oxidation of alcohols in flow.

Scheme 71: Uozumi’s ARP-Pt flow oxidation protocol.

Scheme 72: High-throughput screening of aldehyde oxidation in flow using an in-line GC.

Scheme 73: Permanganate-mediated Nef oxidation of nitroalkanes in flow with the use of in-line sonication to p...

Scheme 74: Continuous-flow aerobic anti-Markovnikov Wacker oxidation.

Scheme 75: Continuous-flow oxidation of 2-benzylpyridine (312) using air as the oxidant.

Scheme 76: Continuous-flow photo-oxygenation of monoterpenes.

Scheme 77: A tubular reactor design for flow photo-oxygenation.

Scheme 78: Glucose oxidase (GOx)-mediated continuous oxidation of glucose using compressed air and the FFMR re...

Scheme 79: Schematic continuous-flow sodium hypochlorite/TEMPO oxidation of alcohols.

Scheme 80: Oxidation using immobilised TEMPO (344) was developed by McQuade et al.

Scheme 81: General protocol for the bleach/catalytic TBAB oxidation of aldehydes and alcohols.

Scheme 82: Continuous-flow PTC-assisted oxidation using hydrogen peroxide. The process was easily scaled up by...

Scheme 83: Continuous-flow epoxidation of cyclohexene (348) and in situ preparation of m-CPBA.

Scheme 84: Continuous-flow epoxidation using DMDO as oxidant.

Scheme 85: Mukayama aerobic epoxidation optimised in flow mode by the Favre-Réguillon group.

Scheme 86: Continuous-flow asymmetric epoxidation of derivatives of 359 exploiting a biomimetic iron catalyst.

Scheme 87: Continuous-flow enzymatic epoxidation of alkenes developed by Watts et al.

Scheme 88: Engineered multichannel microreactor for continuous-flow ozonolysis of 366.

Scheme 89: Continuous-flow synthesis of the vitamin D precursor 368 using multichannel microreactors. MFC: mas...

Scheme 90: Continuous ozonolysis setup used by Kappe et al. for the synthesis of various substrates employing ...

Scheme 91: Continuous-flow apparatus for ozonolysis as developed by Ley et al.

Scheme 92: Continuous-flow ozonolysis for synthesis of vanillin (2) using a film-shear flow reactor.

Scheme 93: Examples of preparative methods for ajoene (386) and allicin (388).

Scheme 94: Continuous-flow oxidation of thioanisole (389) using styrene-based polymer-supported peroxytungstat...

Scheme 95: Continuous oxidation of thiosulfinates using Oxone®-packed reactor.

Scheme 96: Continuous-flow electrochemical oxidation of thioethers.

Scheme 97: Continuous-flow oxidation of 400 to cinnamophenone (235).

Scheme 98: Continuous-flow synthesis of dehydrated material 401 via oxidation of methyl dihydrojasmonate (33).

Scheme 99: Some industrially important transformations involving Grignard reagents.

Scheme 100: Grachev et al. apparatus for continuous preparation of Grignard reagents.

Scheme 101: Example of fluidized Mg bed reactor with NMR spectrometer as on-line monitoring system.

Scheme 102: Continuous-flow synthesis of Grignard reagents and subsequent quenching reaction.

Figure 10: Membrane-based, liquid–liquid separator with integrated pressure control [52]. Adapted with permission ...

Scheme 103: Continuous-flow synthesis of 458, an intermediate to fluconazole (459).

Scheme 104: Continuous-flow synthesis of ketones starting from benzoyl chlorides.

Scheme 105: A Grignard alkylation combining CSTR and PFR technologies with in-line infrared reaction monitoring....

Scheme 106: Continuous-flow preparation of 469 from Grignard addition of methylmagnesium bromide.

Scheme 107: Continuous-flow synthesis of Grignard reagents 471.

Scheme 108: Preparation of the Grignard reagent 471 using CSTR and the continuous process for synthesis of the ...

Scheme 109: Continuous process for carboxylation of Grignard reagents in flow using tube-in-tube technology.

Scheme 110: Continuous synthesis of propargylic alcohols via ethynyl-Grignard reagent.

Scheme 111: Silica-supported catalysed enantioselective arylation of aldehydes using Grignard reagents in flow ...

Scheme 112: Acid-catalysed rearrangement of citral and dehydrolinalool derivatives.

Scheme 113: Continuous stilbene isomerisation with continuous recycling of photoredox catalyst.

Scheme 114: Continuous-flow synthesis of compound 494 as developed by Ley et al.

Scheme 115: Selected industrial applications of DA reaction.

Scheme 116: Multistep flow synthesis of the spirocyclic structure 505 via employing DA cycloaddition.

Scheme 117: Continuous-flow DA reaction developed in a plater flow reactor for the preparation of the adduct 508...

Scheme 118: Continuous-flow DA reaction using a silica-supported imidazolidinone organocatalyst.

Scheme 119: Batch vs flow for the DA reaction of (cyclohexa-1,5-dien-1-yloxy)trimethylsilane (513) with acrylon...

Scheme 120: Continuous-flow DA reaction between 510 and 515 using a shell-core droplet system.

Scheme 121: Continuous-flow synthesis of bicyclic systems from benzyne precursors.

Scheme 122: Continuous-flow synthesis of bicyclic scaffolds 527 and 528 for further development of potential ph...

Scheme 123: Continuous-flow inverse-electron hetero-DA reaction to pyridine derivatives such as 531.

Scheme 124: Comparison between batch and flow for the synthesis of pyrimidinones 532–536 via retro-DA reaction ...

Scheme 125: Continuous-flow coupled with ultrasonic system for preparation of ʟ-ascorbic acid derivatives 539 d...

Scheme 126: Two-step continuous-flow synthesis of triazole 543.

Scheme 127: Continuous-flow preparation of triazoles via CuAAC employing 546-based heterogeneous catalyst.

Scheme 128: Continuous-flow synthesis of compounds 558 through A3-coupling and 560 via AgAAC both employing the...

Scheme 129: Continuous-flow photoinduced [2 + 2] cycloaddition for the preparation of bicyclic derivatives of 5...

Scheme 130: Continuous-flow [2 + 2] and [5 + 2] cycloaddition on large scale employing a flow reactor developed...

Scheme 131: Continuous-flow preparation of the tricyclic structures 573 and 574 starting from pyrrole 570 via [...

Scheme 132: Continuous-flow [2 + 2] photocyclization of cinnamates.

Scheme 133: Continuous-flow preparation of cyclobutane 580 on a 5-plates photoreactor.

Scheme 134: Continuous-flow [2 + 2] photocycloaddition under white LED lamp using heterogeneous PCN as photocat...

Figure 11: Picture of the parallel tube flow reactor (PTFR) "The Firefly" developed by Booker-Milburn et al. a...

Scheme 135: Continuous-flow acid-catalysed [2 + 2] cycloaddition between silyl enol ethers and acrylic esters.

Scheme 136: Continuous synthesis of lactam 602 using glass column reactors.

Scheme 137: In situ generation of ketenes for the Staudinger lactam synthesis developed by Ley and Hafner.

Scheme 138: Application of [2 + 2 + 2] cycloadditions in flow employed by Ley et al.

Scheme 139: Examples of FC reactions applied in F&F industry.

Scheme 140: Continuous-flow synthesis of ibuprofen developed by McQuade et al.

Scheme 141: The FC acylation step of Jamison’s three-step ibuprofen synthesis.

Scheme 142: Synthesis of naphthalene derivative 629 via FC acylation in microreactors.

Scheme 143: Flow system for rapid screening of catalysts and reaction conditions developed by Weber et al.

Scheme 144: Continuous-flow system developed by Buorne, Muller et al. for DSD optimisation of the FC acylation ...

Scheme 145: Continuous-flow FC acylation of alkynes to yield β-chlorovinyl ketones such as 638.

Scheme 146: Continuous-flow synthesis of tonalide (619) developed by Wang et al.

Scheme 147: Continuous-flow preparation of acylated arene such as 290 employing Zr4+-β-zeolite developed by Kob...

Scheme 148: Flow system applied on an Aza-FC reaction catalysed by the thiourea catalyst 648.

Scheme 149: Continuous hydroformylation in scCO2.

Scheme 150: Two-step flow synthesis of aldehyde 655 through a sequential Heck reaction and subsequent hydroform...

Scheme 151: Single-droplet (above) and continuous (below) flow reactors developed by Abolhasani et al. for the ...

Scheme 152: Continuous hydroformylation of 1-dodecene (655) using a PFR-CSTR system developed by Sundmacher et ...

Scheme 153: Continuous-flow synthesis of the aldehyde 660 developed by Eli Lilly & Co. [32]. Adapted with permissio...

Scheme 154: Continuous asymmetric hydroformylation employing heterogenous catalst supported on carbon-based sup...

Scheme 155: Examples of acetylation in F&F industry: synthesis of bornyl (S,R,S-664) and isobornyl (S,S,S-664) ...

Scheme 156: Continuous-flow preparation of bornyl acetate (S,R,S-664) employing the oscillating flow reactor.

Scheme 157: Continuous-flow synthesis of geranyl acetate (666) from acetylation of geraniol (343) developed by ...

Scheme 158: 12-Ttungstosilicic acid-supported silica monolith-catalysed acetylation in flow.

Scheme 159: Continuous-flow preparation of cyclopentenone 676.

Scheme 160: Two-stage synthesis of coumarin (90) via acetylation of salicylaldehyde (88).

Scheme 161: Intensification process for acetylation of 5-methoxytryptamine (677) to melatonin (678) developed b...

Scheme 162: Examples of macrocyclic musky odorants both natural (679–681) and synthetic (682 and 683).

Scheme 163: Flow setup combined with microwave for the synthesis of macrocycle 686 via RCM.

Scheme 164: Continuous synthesis of 2,5-dihydro-1H-pyrroles via ring-closing metathesis.

Scheme 165: Continuous-flow metathesis of 485 developed by Leadbeater et al.

Figure 12: Comparison between RCM performed using different routes for the preparation of 696. On the left the...

Scheme 166: Continuous-flow RCM of 697 employed the solid-supported catalyst 698 developed by Grela, Kirschning...

Scheme 167: Continuous-flow RORCM of cyclooctene employing the silica-absorbed catalyst 700.

Scheme 168: Continuous-flow self-metathesis of methyl oleate (703) employing SILP catalyst 704.

Scheme 169: Flow apparatus for the RCM of 697 using a nanofiltration membrane for the recovery and reuse of the...

Scheme 170: Comparison of loadings between RCMs performed with different routes for the synthesis of 709.
A new and efficient methodology for olefin epoxidation catalyzed by supported cobalt nanoparticles
- Lucía Rossi-Fernández,
- Viviana Dorn and
- Gabriel Radivoy
Beilstein J. Org. Chem. 2021, 17, 519–526, doi:10.3762/bjoc.17.46

- materials and it could be recovered and reused maintaining its unaltered high activity. Keywords: alkenes; cobalt nanoparticles; epoxides; oxidation; TBHP; Introduction Olefin oxidation reactions are key synthetic transformations in the production of oxygenated chemicals of high interest for both academic

Graphical Abstract

Figure 1: TEM micrograph and size distribution graphic for CoNPs@MgO catalyst (scale bar = 20 nm).

Scheme 1: Plausible mechanistic pathway for olefin epoxidation catalyzed by CoNPs/MgO in the presence of t-Bu...
A heterobimetallic tetrahedron from a linear platinum(II)-bis(acetylide) metalloligand
- Matthias Hardy,
- Marianne Engeser and
- Arne Lützen
Beilstein J. Org. Chem. 2020, 16, 2701–2708, doi:10.3762/bjoc.16.220

- amine functions and thus a higher tendency to undergo one-electron oxidation reactions when stored under ambient conditions. In order to check the composition of heterobimetallic 4 we performed mass spectrometric experiments first. Figure 1 shows the ESI mass spectrum of metallosupramolecular cage 4

Graphical Abstract

Scheme 1: Stepwise assembly of the heterobimetallic tetrahedron 4, starting from 4-ethynylaniline (1) and tra...

Figure 1: ESI(+) mass spectrum of heterobimetallic complex 4. The top inset shows the experimentally observed...

Figure 2: UV–vis spectrum of heterobimetallic complex 4 (1150 µM in acetonitrile at 295 K, 0.01 mm cuvette).

Figure 3: Schematic representation of symmetry-considerations concerning possible diastereomeric tetrahedra. ...

Figure 4: Detailed excerpt of the 31P NMR spectrum of 4 (202 MHz, acetonitrile-d3, 298 K).

Figure 5: 1H NMR and DOSY spectrum of heterobimetallic assembly 4 (500 MHz, acetonitilre-d3, 298 K).

Figure 6: GFN2-xTB minimized gas phase models of the cationic units of all possible diastereomers of 4. Color...
Dawn of a new era in industrial photochemistry: the scale-up of micro- and mesostructured photoreactors
- Emine Kayahan,
- Mathias Jacobs,
- Leen Braeken,
- Leen C.J. Thomassen,
- Simon Kuhn,
- Tom van Gerven and
- M. Enis Leblebici
Beilstein J. Org. Chem. 2020, 16, 2484–2504, doi:10.3762/bjoc.16.202

- light path of the reactor. The major drawback of the aerosol photoreactor are safety issues, especially for organic oxidation reactions. Spraying organics into air or oxygen could lead to explosions. Therefore, the lower and upper explosion limits for the several organic photooxidation reactions should

Graphical Abstract

Figure 1: The momentum transport affects the mass transfer and the light field. All transport phenomena need ...

Figure 2: Common photomicroreactor designs: (a) Straight channel, (b) serpentine channel, (c) square serpenti...

Figure 3: Benchmarked photoreactors: (a) Microcapillaries in parallel, (b) microcapillaries in series, (c) fl...

Figure 4: Photochemical reactions that are detailed in Table 1.

Figure 5: Structured reactors designed for enhancing the mass transfer: (a) Packed bed photoreactor, (b) mono...

Figure 6: Comparison of the LED board designs of photomicroreactors: (a) CC array design, (b) MC array design...

Figure 7: Illustration of the light scattering phenomenon inside a photocatalytic flow reactor.

Figure 8: Efficiency of the absorption process in scattering situations with respect to pure absorption situa...

Figure 9: Different types of distributors: (a) Traditional or consecutive manifold, (b) bifurcation unit dist...
An overview on disulfide-catalyzed and -cocatalyzed photoreactions
- Yeersen Patehebieke
Beilstein J. Org. Chem. 2020, 16, 1418–1435, doi:10.3762/bjoc.16.118

- cyclization, and an elimination give the desired product 27 (Scheme 7). The use of PhSSPh obviated the undesired reactions, which occurred when using Ph3SnH and Bu3SnH, respectively, as the catalyst for this reaction. Oxidation reactions Not only can disulfide catalysts induce cycloadditions of olefins but

Graphical Abstract

Scheme 1: [3 + 2] cyclization catalyzed by diaryl disulfide.

Scheme 2: [3 + 2] cycloaddition catalyzed by disulfide.

Scheme 3: Disulfide-bridged peptide-catalyzed enantioselective cycloaddition.

Scheme 4: Disulfide-catalyzed [3 + 2] methylenecyclopentane annulations.

Scheme 5: Disulfide as a HAT cocatalyst in the [4 + 2] cycloaddition reaction.

Scheme 6: Proposed mechanism of the [4 + 2] cycloaddition reaction using disulfide as a HAT cocatalyst.

Scheme 7: Disulfide-catalyzed ring expansion of vinyl spiro epoxides.

Scheme 8: Disulfide-catalyzed aerobic oxidation of diarylacetylene.

Scheme 9: Disulfide-catalyzed aerobic photooxidative cleavage of olefins.

Scheme 10: Disulfide-catalyzed aerobic oxidation of 1,3-dicarbonyl compounds.

Scheme 11: Proposed mechanism of the disulfide-catalyzed aerobic oxidation of 1,3-dicarbonyl compounds.

Scheme 12: Disulfide-catalyzed oxidation of allyl alcohols.

Scheme 13: Disulfide-catalyzed diboration of alkynes.

Scheme 14: Dehalogenative radical cyclization catalyzed by disulfide.

Scheme 15: Hydrodifluoroacetamidation of alkenes catalyzed by disulfide.

Scheme 16: Plausible mechanism of the hydrodifluoroacetamidation of alkenes catalyzed by disulfide.

Scheme 17: Disulfide-cocatalyzed anti-Markovnikov olefin hydration reactions.

Scheme 18: Disulfide-catalyzed decarboxylation of carboxylic acids.

Scheme 19: Proposed mechanism of the disulfide-catalyzed decarboxylation of carboxylic acids.

Scheme 20: Disulfide-catalyzed decarboxylation of carboxylic acids.

Scheme 21: Disulfide-catalyzed conversion of maleate esters to fumarates and 5H-furanones.

Scheme 22: Disulfide-catalyzed isomerization of difluorotriethylsilylethylene.

Scheme 23: Disulfide-catalyzed isomerization of allyl alcohols to carbonyl compounds.

Scheme 24: Proposed mechanism for the disulfide-catalyzed isomerization of allyl alcohols to carbonyl compound...

Scheme 25: Diphenyl disulfide-catalyzed enantioselective synthesis of ophirin B.

Scheme 26: Disulfide-catalyzed isomerization in the total synthesis of (+)-hitachimycin.

Scheme 27: Disulfide-catalyzed isomerization in the synthesis of (−)-gloeosporone.
Synthesis of 3-substituted isoxazolidin-4-ols using hydroboration–oxidation reactions of 4,5-unsubstituted 2,3-dihydroisoxazoles
- Lívia Dikošová,
- Júlia Laceková,
- Ondrej Záborský and
- Róbert Fischer
Beilstein J. Org. Chem. 2020, 16, 1313–1319, doi:10.3762/bjoc.16.112

Graphical Abstract

Figure 1: 3-Substituted isoxazolidin-4-ols resembling 3-hydroxypyrrolidines.

Scheme 1: Synthetic approach towards isoxazolidin-4-ols via the regioselective reductive cleavage of the C5–O...

Scheme 2: Hydroboration-oxidation of 4,5-unsubstituted 2,3-dihydroisoxazoles.

Figure 2: Selected NOE enhancements observed in the isoxazolidin-4-ol trans-8a. The arrows show the NOESY cor...

Scheme 3: Dess-Martin oxidation of isoxazolidin-4-ols to ketones.

Scheme 4: Inversion of the relative configuration of the isoxazolidine ring.

Figure 3: Selected NOE enhancements observed in the isoxazolidin-4-ol cis-10a. The arrows show the NOESY corr...

Scheme 5: N-debenzylation via N-Troc-protected isoxazolidines.
Copper catalysis with redox-active ligands
- Agnideep Das,
- Yufeng Ren,
- Cheriehan Hessin and
- Marine Desage-El Murr
Beilstein J. Org. Chem. 2020, 16, 858–870, doi:10.3762/bjoc.16.77

- copper intermediates in oxidation reactions in biological systems is very well documented [9]. It is therefore not surprising that this would be an area of choice for the development of bioinspired and/or redox-active copper complexes. Inspired by the pioneering works by Stack [10], Wieghardt and

Graphical Abstract

Scheme 1: Copper complexes with amidophenolate type benzoxazole ligands for alcohol oxidations.

Scheme 2: Copper-catalyzed aerobic oxidation of alcohols and representative substrate scope.

Scheme 3: Introduction of H-bonding network in the ligand coordination sphere.

Scheme 4: Well-defined isatin copper complexes.

Scheme 5: Catalyst control in the biomimetic phenol ortho-oxidation.

Scheme 6: Structural diversity accessible by direct functionalization.

Scheme 7: Copper-catalyzed trifluoromethylation of heteroaromatics with redox-active iminosemiquinone ligands....

Scheme 8: Reversal of helical chirality upon redox stimuli and enantioselective Michael addition with a redox...

Scheme 9: Interaction of guanidine-copper catalyst with oxygen and representative coupling products. a4 mol %...

Scheme 10: Access to 1,2-oxy-aminoarenes by copper-catalyzed phenol–amine coupling.

Scheme 11: Copper-catalyzed aziridination through molecular spin catalysis with redox-active iminosemiquinone ...

Scheme 12: Nitrogen-group and carbon-group transfer in copper-catalyzed aziridination and cyclopropanation thr...
A systematic review on silica-, carbon-, and magnetic materials-supported copper species as efficient heterogeneous nanocatalysts in “click” reactions
- Pezhman Shiri and
- Jasem Aboonajmi
Beilstein J. Org. Chem. 2020, 16, 551–586, doi:10.3762/bjoc.16.52
- delivery [93], and cancer detection [94]. In this regard, various functionalized MNPs were employed in diverse organic reactions, including Suzuki–Miyaura reactions, Sonogashira reactions, Ullmann coupling, hydrogenation reactions, oxidation reactions, hydroformylation reactions, and “click” reactions [89

Graphical Abstract

Scheme 1: Chemical structure of the catalysts 1a and 1b and their catalytic application in CuAAC reactions.

Scheme 2: Synthetic route to the catalyst 11 and its catalytic application in CuAAC reactions.

Scheme 3: Synthetic route of dendrons, illustrated using G2-AMP 23.

Scheme 4: The catalytic application of CuYAu–Gx-AAA–SBA-15 in a CuAAC reaction.

Scheme 5: Synthetic route to the catalyst 36.

Scheme 6: Application of the catalyst 36 in CuAAC reactions.

Scheme 7: The synthetic route to the catalyst 45 and catalytic application of 45 in “click” reactions.

Scheme 8: Synthetic route to the catalyst 48 and catalytic application of 48 in “click” reactions.

Scheme 9: Synthetic route to the catalyst 58 and catalytic application of 58 in “click” reactions.

Scheme 10: Synthetic route to the catalyst 64 and catalytic application of 64 in “click” reactions.

Scheme 11: Chemical structure of the catalyst 68 and catalytic application of 68 in “click” reactions.

Scheme 12: Chemical structure of the catalyst 69 and catalytic application of 69 in “click” reactions.

Scheme 13: Synthetic route to, and chemical structure of the catalyst 74.

Scheme 14: Application of the cayalyst 74 in “click” reactions.

Scheme 15: Synthetic route to, and chemical structure of the catalyst 78 and catalytic application of 78 in “c...

Scheme 16: Synthetic route to the catalyst 85.

Scheme 17: Application of the catalyst 85 in “click” reactions.

Scheme 18: Synthetic route to the catalyst 87 and catalytic application of 87 in “click” reactions.

Scheme 19: Chemical structure of the catalyst 88 and catalytic application of 88 in “click” reactions.

Scheme 20: Synthetic route to the catalyst 90 and catalytic application of 90 in “click” reactions.

Scheme 21: Synthetic route to the catalyst 96 and catalytic application of 96 in “click” reactions.

Scheme 22: Synthetic route to the catalyst 100 and catalytic application of 100 in “click” reactions.

Scheme 23: Synthetic route to the catalyst 102 and catalytic application of 23 in “click” reactions.

Scheme 24: Synthetic route to the catalysts 108–111.

Scheme 25: Catalytic application of 108–111 in “click” reactions.

Scheme 26: Synthetic route to the catalyst 121 and catalytic application of 121 in “click” reactions.

Scheme 27: Synthetic route to 125 and application of 125 in “click” reactions.

Scheme 28: Synthetic route to the catalyst 131 and catalytic application of 131 in “click” reactions.

Scheme 29: Synthetic route to the catalyst 136.

Scheme 30: Application of the catalyst 136 in “click” reactions.

Scheme 31: Synthetic route to the catalyst 141 and catalytic application of 141 in “click” reactions.

Scheme 32: Synthetic route to the catalyst 144 and catalytic application of 144 in “click” reactions.

Scheme 33: Synthetic route to the catalyst 149 and catalytic application of 149 in “click” reactions.

Scheme 34: Synthetic route to the catalyst 153 and catalytic application of 153 in “click” reactions.

Scheme 35: Synthetic route to the catalyst 155 and catalytic application of 155 in “click” reactions.

Scheme 36: Synthetic route to the catalyst 157 and catalytic application of 157 in “click” reactions.

Scheme 37: Synthetic route to the catalyst 162.

Scheme 38: Application of the catalyst 162 in “click” reactions.

Scheme 39: Synthetic route to the catalyst 167 and catalytic application of 167 in “click” reactions.

Scheme 40: Synthetic route to the catalyst 169 and catalytic application of 169 in “click” reactions.

Scheme 41: Synthetic route to the catalyst 172.

Scheme 42: Application of the catalyst 172 in “click” reactions.
Recent advances in photocatalyzed reactions using well-defined copper(I) complexes
- Mingbing Zhong,
- Xavier Pannecoucke,
- Philippe Jubault and
- Thomas Poisson
Beilstein J. Org. Chem. 2020, 16, 451–481, doi:10.3762/bjoc.16.42
- addition of a trifluoromethyl radical to silyl enol ethers derived from ketones using the same reaction conditions (Scheme 12) [28]. 1.3 Oxidation reactions In 2015, Bissember and co-workers used the Sauvage catalyst to generate an α-amino radical, which was used to perform the synthesis of annulated
- the presence of the acid, it is protonated and collapses into the corresponding alkoxy radical. This radical carries out a 1,5-HAT to generate a carbon-centered radical. Finally, this radical recombines with the α-amino radical generated from the above-mentioned pathway. 2.3 Oxidation reactions In

Graphical Abstract

Scheme 1: [Cu(I)(dap)2]Cl-catalyzed ATRA reaction under green light irradiation.

Scheme 2: Photocatalytic allylation of α-haloketones.

Scheme 3: [Cu(I)(dap)2]Cl-photocatalyzed chlorosulfonylation and chlorotrifluoromethylation of alkenes.

Scheme 4: Photocatalytic perfluoroalkylchlorination of electron-deficient alkenes using the Sauvage catalyst.

Scheme 5: Photocatalytic synthesis of fluorinated sultones.

Scheme 6: Photocatalyzed haloperfluoroalkylation of alkenes and alkynes.

Scheme 7: Chlorosulfonylation of alkenes catalyzed by [Cu(I)(dap)2]Cl. aNo Na2CO3 was added. b1 equiv of Na2CO...

Scheme 8: Copper-photocatalyzed reductive allylation of diaryliodonium salts.

Scheme 9: Copper-photocatalyzed azidomethoxylation of olefins.

Scheme 10: Benzylic azidation initiated by [Cu(I)(dap)2]Cl.

Scheme 11: Trifluoromethyl methoxylation of styryl derivatives using [Cu(I)(dap)2]PF6. All redox potentials ar...

Scheme 12: Trifluoromethylation of silyl enol ethers.

Scheme 13: Synthesis of annulated heterocycles upon oxidation with the Sauvage catalyst.

Scheme 14: Oxoazidation of styrene derivatives using [Cu(dap)2]Cl as a precatalyst.

Scheme 15: [Cu(I)(dpp)(binc)]PF6-catalyzed ATRA reaction.

Scheme 16: Allylation reaction of α-bromomalonate catalyzed by [Cu(I)(dpp)(binc)]PF6 following an ATRA mechani...

Scheme 17: Bromo/tribromomethylation reaction using [Cu(I)(dmp)(BINAP)]PF6.

Scheme 18: Chlorotrifluoromethylation of alkenes catalyzed by [Cu(I)(N^N)(xantphos)]PF6.

Scheme 19: Chlorosulfonylation of styrene and alkyne derivatives by ATRA reactions.

Scheme 20: Reduction of aryl and alkyl halides with the complex [Cu(I)(bcp)(DPEPhos)]PF6. aIrradiation was car...

Scheme 21: Meerwein arylation of electron-rich aromatic derivatives and 5-exo-trig cyclization catalyzed by th...

Scheme 22: [Cu(I)(bcp)(DPEPhos)]PF6-photocatalyzed synthesis of alkaloids. aYield over two steps (cyclization ...

Scheme 23: Copper-photocatalyzed decarboxylative amination of NHP esters.

Scheme 24: Photocatalytic decarboxylative alkynylation using [Cu(I)(dq)(binap)]BF4.

Scheme 25: Copper-photocatalyzed alkylation of glycine esters.

Scheme 26: Copper-photocatalyzed borylation of organic halides. aUnder continuous flow conditions.

Scheme 27: Copper-photocatalyzed α-functionalization of alcohols with glycine ester derivatives.

Scheme 28: δ-Functionalization of alcohols using [Cu(I)(dmp)(xantphos)]BF4.

Scheme 29: Photocatalytic synthesis of [5]helicene and phenanthrene.

Scheme 30: Oxidative carbazole synthesis using in situ-formed [Cu(I)(dmp)(xantphos)]BF4.

Scheme 31: Copper-photocatalyzed functionalization of N-aryl tetrahydroisoquinolines.

Scheme 32: Bicyclic lactone synthesis using a copper-photocatalyzed PCET reaction.

Scheme 33: Photocatalytic Pinacol coupling reaction catalyzed by [Cu(I)(pypzs)(BINAP)]BF4. The ligands of the ...

Scheme 34: Azide photosensitization using a Cu-based photocatalyst.
A review of asymmetric synthetic organic electrochemistry and electrocatalysis: concepts, applications, recent developments and future directions
- Munmun Ghosh,
- Valmik S. Shinde and
- Magnus Rueping
Beilstein J. Org. Chem. 2019, 15, 2710–2746, doi:10.3762/bjoc.15.264

- chirally modified electrodes Electrochemical oxidation reactions have long served as substantial synthetic tool because of their ability to increase the functionality of organic molecules via reversing the polarity of electron-rich functional groups and thereby generating highly reactive intermediates
- be considered indirect electrolysis as the mediator takes an electron from the solid electrode and performs the organic transformation. This strategy has been shown to be suitable for inducing chirality, particularly in electro-oxidation reactions. Many chiral TEMPO-derived compounds have been

Graphical Abstract

Figure 1: General classification of asymmetric electroorganic reactions.

Scheme 1: Asymmetric reduction of 4-acetylpyridine using a modified graphite cathode.

Scheme 2: Asymmetric hydrogenation of ketones using Raney nickel powder electrodes modified with optically ac...

Scheme 3: Asymmetric reduction of prochiral activated olefins with a poly-ʟ-valine-coated graphite cathode.

Scheme 4: Asymmetric reduction of prochiral carbonyl compounds, oximes and gem-dibromides on a poly-ʟ-valine-...

Scheme 5: Asymmetric hydrogenation of prochiral ketones with poly[RuIII(L)2Cl2]+-modified carbon felt cathode...

Scheme 6: Asymmetric hydrogenation of α-keto esters using chiral polypyrrole film-coated cathode incorporated...

Scheme 7: Quinidine and cinchonidine alkaloid-induced asymmetric electroreduction of acetophenone.

Scheme 8: Asymmetric electroreduction of 4- and 2-acetylpyridines at a mercury cathode in the presence of a c...

Scheme 9: Enantioselective reduction of 4-methylcoumarin in the presence of catalytic yohimbine.

Scheme 10: Cinchonine-induced asymmetric electrocarboxylation of 4-methylpropiophenone.

Scheme 11: Enantioselective hydrogenation of methyl benzoylformate using an alkaloid entrapped silver cathode.

Scheme 12: Alkaloid-induced enantioselective hydrogenation using a Cu nanoparticle cathode.

Scheme 13: Alkaloid-induced enantioselective hydrogenation of aromatic ketones using a bimetallic Pt@Cu cathod...

Scheme 14: Enantioselective reduction of ketones at mercury cathode using N,N'-dimethylquininium tetrafluorobo...

Scheme 15: Asymmetric synthesis of an amino acid using an electrode modified with amino acid oxidase and elect...

Scheme 16: Asymmetric oxidation of p-tolyl methyl sulfide using chemically modified graphite anode.

Scheme 17: Asymmetric oxidation of unsymmetric sulfides using poly(amino acid)-coated electrodes.

Scheme 18: Enantioselective, electocatalytic oxidative coupling on TEMPO-modified graphite felt electrode in t...

Scheme 19: Asymmetric electrocatalytic oxidation of racemic alcohols on a TEMPO-modified graphite felt electro...

Scheme 20: Asymmetric electrocatalytic lactonization of diols on TEMPO-modified graphite felt electrodes.

Scheme 21: Asymmetric electrochemical pinacolization in a chiral solvent.

Scheme 22: Asymmetric electroreduction using a chiral supporting electrolyte.

Scheme 23: Asymmetric anodic oxidation of enol acetates using chiral supporting electrolytes.

Scheme 24: Kinetic resolution of primary amines using a chiral N-oxyl radical mediator.

Scheme 25: Chiral N-oxyl-radical-mediated kinetic resolution of secondary alcohols via electrochemical oxidati...

Scheme 26: Chiral iodoarene-mediated asymmetric electrochemical lactonization.

Scheme 27: Os-catalyzed electrochemical asymmetric dihydroxylation of olefins using the Sharpless ligand and i...

Scheme 28: Asymmetric electrochemical epoxidation of olefins catalyzed by a chiral Mn-salen complex.

Scheme 29: Asymmetric electrooxidation of 1,2-diols, and amino alcohols using a chiral copper catalyst.

Scheme 30: Mechanism of asymmetric electrooxidation of 1,2-diols, and amino alcohols using a chiral copper cat...

Scheme 31: Enantioselective electrocarboxylation catalyzed by an electrogenerated chiral [CoI(salen)]− complex....

Scheme 32: Asymmetric oxidative cross coupling of 2-acylimidazoles with silyl enol ethers.

Scheme 33: Ni-catalyzed asymmetric electroreductive cleavage of allylic β-keto ester 89.

Scheme 34: Asymmetric alkylation using a combination of electrosynthesis and a chiral Ni catalyst.

Scheme 35: Mechanism of asymmetric alkylation using a combination of electrosynthesis and a chiral Ni catalyst....

Scheme 36: Asymmetric epoxidation by electrogenerated percarbonate and persulfate ions in the presence of chir...

Scheme 37: α-Oxyamination of aldehydes via anodic oxidation catalyzed by chiral secondary amines.

Scheme 38: The α-alkylation of aldehydes via anodic oxidation catalyzed by chiral secondary amines.

Scheme 39: Mechanism of α-alkylation of aldehydes via anodic oxidation catalyzed by chiral secondary amines.

Scheme 40: Electrochemical chiral secondary amine-catalyzed intermolecular α-arylation of aldehydes.

Scheme 41: Mechanism of electrochemical chiral secondary amine-catalyzed intermolecular α-arylation of aldehyd...

Scheme 42: Asymmetric cross-dehydrogenative coupling of tertiary amines with simple ketones via an electrochem...

Scheme 43: Electroenzymatic asymmetric reduction using enoate reductase.

Scheme 44: Assymetric reduction using alcohol dehydrogenase as the electrocatalyst.

Scheme 45: Asymmetric electroreduction catalyzed by thermophilic NAD-dependent alcohol dehydrogenase.

Scheme 46: Asymmetric epoxidation of styrene by electrochemical regeneration of flavin-dependent monooxygenase....

Scheme 47: Asymmetric electroreduction using a chloroperoxidase catalyst.

Scheme 48: Asymmetric electrochemical transformation mediated by hydrophobic vitamin B12.

Scheme 49: Diastereoselective cathodic reduction of phenylglyoxalic acids substituted with amines as chiral au...

Scheme 50: Ni-catalyzed asymmetric electroreductive cross coupling of aryl halides with α-chloropropanoic acid...

Scheme 51: Electrochemical Mannich addition of silyloxyfuran to in situ-generated N-acyliminium ions.

Scheme 52: Stereoselective electroreductive homodimerization of cinnamates attached to a camphor-derived chira...

Scheme 53: Diastereoselective electrochemical carboxylation of chiral α-bromocarboxylic acid derivatives.

Scheme 54: Electrocatalytic stereoselective conjugate addition of chiral β-dicarbonyl compounds to methyl viny...

Scheme 55: Stereoselective electrochemical carboxylation of chiral cinnamic acid derivatives under a CO2 atmos...

Scheme 56: Electrochemical diastereoselective α-alkylation of pyrrolidines attached with phosphorus-derived ch...

Scheme 57: Electrogenerated cyanomethyl anion-induced synthesis of chiral cis-β-lactams from amides bearing ch...

Scheme 58: Diastereoselective anodic oxidation followed by intramolecular cyclization of ω-hydroxyl amides bea...

Scheme 59: Electrochemical deprotonation of Ni(II) glycinate containing (S)-BPB as a chiral auxiliary: diaster...

Scheme 60: Enantioselective electroreductive coupling of diaryl ketones with α,β-unsaturated carbonyl compound...

Scheme 61: Asymmetric total synthesis of ropivacaine and its analogues using a electroorganic reaction as a ke...

Scheme 62: Asymmetric total synthesis of (−)-crispine A and its natural enantiomer via anodic cyanation of tet...

Scheme 63: Asymmetric oxidative electrodimerization of cinnamic acid derivatives as key step for the synthesis...
Synthetic terpenoids in the world of fragrances: Iso E Super® is the showcase
- Alexey Stepanyuk and
- Andreas Kirschning
Beilstein J. Org. Chem. 2019, 15, 2590–2602, doi:10.3762/bjoc.15.252
- process is commonly associated with oxidation reactions, while the latter process is often based on the action of lipases. Very recently, a new concept was disclosed that probed sesquiterpene cyclases to accept unnatural farnesyl pyrophosphates and generate unnatural cyclisation products with unusual

Graphical Abstract

Figure 1: Terpene constituents 1–9 found in geranium and bergamot oils and specified odours of individual com...

Figure 2: Other selected mono- and sesquiterpenes (10–26) as fragrance materials [6].

Figure 3: Main constituents of natural iris oil: irone (27).

Scheme 1: First synthesis of ionone (30) [11].

Scheme 2: First synthesis of Ambrelux (32) [14].

Scheme 3: Industrial synthesis of myrcene (1) by pyrolysis of β-pinene (8).

Scheme 4: First synthesis of Iso E Super® (33), Iso E Super Plus® (34) and Georgywood® (35) as a mixture of i...

Figure 4: Iso E Super® region of GC spectra of Molecule 01 (left, 75 €–100 € per 100 mL; march 2019), a low-p...

Scheme 5: First synthetic route to (−)-Georgywood® (35) by Corey and Hong [33].

Scheme 6: First synthetic route to the odour-active (+)-enantiomer of Iso E Super Plus® (+)-34 [33].

Scheme 7: Analysis of the isomerisation process and formation of products. Most importantly, Iso E Super® (33...

Scheme 8: Isomerisation using additives such as alcohols or carboxylic acids. The product with the γ-position...

Scheme 9: Iso E Super Plus® (34) can undergo a third cyclisation to tetrahydrofuran 59 through compound rac-53...

Figure 5: (Adapted from ref. [8]) Ionone (30, 1893, odour threshold: 0.8 ng L−1), koavone (1982, odour threshold...

Figure 6: Branched, terpene-like cyclohexene derivatives, that are synthetic fragrance components: 60: Iso da...

Scheme 10: New unnatural terpenoid 70 from unnatural farnesyl pyrophosphate derivative 69 and comparison with ...
Metal-free mechanochemical oxidations in Ertalyte® jars
- Andrea Porcheddu,
- Francesco Delogu,
- Lidia De Luca,
- Claudia Fattuoni and
- Evelina Colacino
Beilstein J. Org. Chem. 2019, 15, 1786–1794, doi:10.3762/bjoc.15.172

- . In particular, in this study, we used sodium hypochlorite pentahydrate (NaOCl·5H2O) in the presence of a catalytic amount of a nitrosyl radical (TEMPO or AZADO) to induce mechanochemical oxidation reactions on suitably selected primary and secondary alcohols. Performed in a high-energy ball mill and
- -assisted oxidation reactions and, we have gained valuable experience in handling this reagent in several mechanochemical applications [45][46]. N-Chlorosuccinimide (1.1 mmol) and 3-pheny-1-propanol (1.0 mmol) were milled together in the presence of TEMPO (5 mol %), K2CO3 (4.0 mmol) and KBr (3.0 mol %) for

Graphical Abstract

Scheme 1: Oxidation of 3-pheny-1-propanol (1a) with N-chlorosuccinimide (NCS) in the presence of (2,2,6,6-tet...

Scheme 2: Hypothesized pathways for the TEMPO-assisted oxidation of alcohols in a) basic or b) acidic reactio...

Scheme 3: TEMPO-assisted oxidation of 3-pheny-1-propanol (1a) under mechanical activation conditions. aPercen...

Scheme 4: Scope of primary alcohol oxidation under mechanical activation conditions. aAll yields refer to iso...

Scheme 5: Proposed mechanism for the oxidation of benzylic alcohols 6a and 7a under mechanochemical condition...

Scheme 6: Scope of secondary alcohols in the oxidation under mechanical activation conditions. aAll yields re...

Scheme 7: Possible mechanism for the TEMPO-mediated oxidation of primary and secondary alcohols by using NaOC...
Transient and intermediate carbocations in ruthenium tetroxide oxidation of saturated rings
- Manuel Pedrón,
- Laura Legnani,
- Maria-Assunta Chiacchio,
- Pierluigi Caramella,
- Tomás Tejero and
- Pedro Merino
Beilstein J. Org. Chem. 2019, 15, 1552–1562, doi:10.3762/bjoc.15.158

- considering solvent effects for acetonitrile and water and their corresponding energy values were calculated at the same level. Since the experimental conditions for the oxidation reactions usually involve a polar medium containing water, all discussions were based on data obtained considering solvent effects

Graphical Abstract

Scheme 1: Oxidation of alkanes with RuO4.

Scheme 2: Mechanisms for RuO4 oxidation of alkanes.

Scheme 3: Oxidation of saturated five-membered (hetero)cyclic compounds.

Scheme 4: Rate-limiting step for the oxidation of cyclopentane (R1), tetrahydrofuran (R2) and tetrahydrothiop...

Figure 1: Optimized (B3LYP-d3bj/Def2SVP/cpcm=MeCN) geometries of transition structures corresponding to the o...

Figure 2: ELF analysis for the oxidation of cyclopentane (R1). Left: evolution of the electron population alo...

Figure 3: ELF analysis for the oxidation of tetrahydrofuran (R2, A) and tetrahydrothiophene (R3, B). Left: ev...

Figure 4: ELF assignment of electrons to the Ru environment. C(Ru) corresponds to a monosynaptic core basin a...

Scheme 5: Rate-limiting step for the oxidation of N-methyl- and N-benzylpyrrolidines R4 and R5, respectively.

Figure 5: Energy profile for the oxidation of R4 and R5. Relative energies, calculated at the B3LYP-d3bj/Def2...

Figure 6: Optimized (B3LYP-d3bj/Def2SVP/cpcm=water) transition structures for the oxidation of R4 and R5.
Synthesis of pyrrolo[1,2-a]quinolines by formal 1,3-dipolar cycloaddition reactions of quinolinium salts
- Anthony Choi,
- Rebecca M. Morley and
- Iain Coldham
Beilstein J. Org. Chem. 2019, 15, 1480–1484, doi:10.3762/bjoc.15.149
- could be converted to other derivatives by Suzuki–Miyaura coupling, reduction or oxidation reactions. Keywords: azomethine ylide; cycloaddition; heterocycle; pyrrolidine; stereoselective; Introduction Cycloaddition reactions of azomethine ylides are an important class of pericyclic reactions that give

Graphical Abstract

Scheme 1: Reaction of ketone 1 with electron-deficient alkenes 2.

Scheme 2: Reactions of ester 4 and amide 5 with electron-deficient alkenes 6.

Figure 1: Single crystal X-ray structure for 7c.

Scheme 3: Reactions of ester 4 and amide 5 with N-methylmaleimide.

Figure 2: Single crystal X-ray structure for 9.

Scheme 4: Reduction and oxidation of adducts 9 and 10.

Scheme 5: Formation of amides 15a and 15b and Suzuki–Miyaura coupling to yield 16.
Synthesis of polydicyclopentadiene using the Cp2TiCl2/Et2AlCl catalytic system and thin-layer oxidation of the polymer in air
- Zhargolma B. Bazarova,
- Ludmila S. Soroka,
- Alex A. Lyapkov,
- Мekhman S. Yusubov and
- Francis Verpoort
Beilstein J. Org. Chem. 2019, 15, 733–745, doi:10.3762/bjoc.15.69

- proceeding polymer oxidation reactions. However, the resulting film of oxidized crosslinked polydicyclopentadiene prevents further penetration of oxygen into the depth of the polymer layer (Figure 15). At this stage in general, the oxidation process is limited by the diffusion of oxygen in the thickness of

Graphical Abstract

Figure 1: Absorption spectra in the UV and visible spectral region: 1) bis(cyclopentadienyl)titan dichloride (...

Figure 2: Absorption spectra in the visible spectral region: 1) Cp2TiCl2·AlEt2Cl (toluene, 10 mmol/L, Ti/Al r...

Figure 3: 1Н NMR spectra of tricyclopentadiene (a) and the interaction product between Cp2TiCl2 and AlEt2Cl w...

Scheme 1: Mechanism of alkylation of Cp2TiCl2.

Figure 4: Visible spectra of a mixture of Cp2TiCl2 and AlEt2Cl as function of time.

Figure 5: Thermometric curve of DCPD polymerization using the catalyst system based on Cp2TiCl2 (a) and its s...

Scheme 2: The structures formed as a result of the cationic polymerization of dicyclopentadiene.

Scheme 3: The units resulting from ROMP of dicyclopentadiene.

Scheme 4: Mechanism of ROMP dicyclopentadiene.

Figure 6: FTIR spectrum of PDCPD obtained in toluene with the catalyst system based on Cp2TiCl2 and AlEt2Cl.

Figure 7: 1Н NMR spectrum of PDCPD obtained with the catalytic system based on Cp2TiCl2 and AlEt2Cl.

Figure 8: GPC traces for two samples of DCPD polymers obtained at a concentration of Cp2TiCl2/AlEt2Cl complex...

Figure 9: IR spectra of cationic polymerized dicyclopentadiene taken after certain periods of time exposed to...

Figure 10: Correlation of intensities of vibrational bands at 1620 and 700 cm−1 and layer exposure time in air...

Figure 11: DSC exotherm for PDCPD subjected to air oxidation for 700 hours.

Figure 12: DSC exotherm for PDCPD subjected to unexposed film: 1) in air atmosphere; 2) in argon.

Scheme 5: Possible radical formation in the reaction (1).

Scheme 6: The first step of the chain propagation.

Figure 13: Dependence of intensities of adsorption bands at 1410 and 700 cm−1 and dwell time of the layer in a...

Figure 14: Semi-logarithmic kinetic curve of PDCPD oxidation in air (thin layer on silicon) with respect to in...

Figure 15: The distribution of oxygen concentration in the polymer layer: 1 – a layer of oxidized cross-linked...

Figure 16: Dependence of the ratio of adsorption bands at 1700 and 700 cm−1 on the exposure time of the layer ...

Figure 17: Infrared spectra (a) of products of cationic polymerization of DCPD, stabilized with an antioxidant...
The LANCA three-component reaction to highly substituted β-ketoenamides – versatile intermediates for the synthesis of functionalized pyridine, pyrimidine, oxazole and quinoxaline derivatives
- Tilman Lechel,
- Roopender Kumar,
- Mrinal K. Bera,
- Reinhold Zimmer and
- Hans-Ulrich Reissig
Beilstein J. Org. Chem. 2019, 15, 655–678, doi:10.3762/bjoc.15.61
- derivatives PM allows a variety of subsequent transformations to new derivatives. The C-4 methyl (in one case benzyl) group is an inevitable structural feature of these pyrimidines, but it can smoothly be used for oxidation reactions to introduce new functional groups. As typical examples selenium dioxide
- with a nonafloxy group all kinds of palladium-catalyzed coupling reactions. Specific oxidation reactions also lead to a variety of new heterocyclic compounds. All the examples collected here show the potential of this approach to highly functionalized heterocycles, furnishing compounds with a very high

Graphical Abstract

Scheme 1: Discovery of the LANCA three-component reaction. The reaction of pivalonitrile (1) with lithiated m...

Scheme 2: Proposed mechanism of the LANCA three-component reaction to β-ketoenamides KE and pyridin-4-ol deri...

Scheme 3: One-pot preparation of pyridin-4-ols PY and their subsequent transformations to highly substituted ...

Scheme 4: Synthesis of β-ketoenamides KE by the LANCA three-component reaction of alkoxyallenes, nitriles and...

Scheme 5: β-Ketoenamides KE36–43 derived from enantiopure components.

Scheme 6: Bis-β-ketoenamides KE44–46 derived from aromatic dicarboxylic acids.

Scheme 7: Conversion of alkyl propargyl ethers E into aryl-substituted β-ketoenamides KEAr and selected produ...

Scheme 8: Condensation of LANCA-derived β-ketoenamides KE with ammonium salts to give 5-alkoxy-substituted py...

Scheme 9: Synthesis of PM31–35 from β-ketoenamides KE37, KE38, KE40, KE41 and KE78 obtained by method A (NH4O...

Scheme 10: Synthesis of bis-pyrimidine derivatives PM36, PM39 and PM40 from β-ketoenamides KE44–46 by method A...

Scheme 11: Functionalization of pyrimidine derivatives PM through selenium dioxide oxidations of PM5, PM9, PM15...

Scheme 12: Conversion of 2-vinyl-substituted pyrimidine PM7 into aldehyde PM50; (NMO = N-methylmorpholine N-ox...

Scheme 13: Deprotection of 5-alkoxy-substituted pyrimidines PM2, PM20 and PM29 and conversion into nonaflates ...

Scheme 14: Palladium-catalyzed coupling reactions of PM54 and PM12 giving rise to new pyrimidine derivatives P...

Scheme 15: Synthesis of pyrimidyl-substituted pyridyl nonaflate PM60.

Scheme 16: Condensation of LANCA-derived β-ketoenamides KE with hydroxylamine hydrochloride leading to pyrimid...

Scheme 17: Reactions of β-ketoenamides KE15 and KE7 with hydroxylamine hydrochloride leading to pyrimidine N-o...

Scheme 18: Structures of pyrimidine N-oxides PO30–33 derived from β-ketoenamides KE43, KE45, KE78 and KE80.

Scheme 19: Reduction of PO4 to PM5 and Boekelheide rearrangements of PO13, PO14, PO4 and PO30 to 4-acetoxymeth...

Scheme 20: Deprotection of 4-acetoxymethyl-substituted pyrimidine derivatives PM61 and PM63, oxidations to for...

Scheme 21: Synthesis of pyrimidinyl-substituted alkyne PM74 and conversion into furopyrimidine PM75 and Sonoga...

Scheme 22: Trifluoroacetic acid-promoted conversion of LANCA-derived β-ketoenamides KE into oxazoles OX and 1,...

Scheme 23: Conversion of β-ketoenamide KE79 into oxazole OX16 and transformation into 5-styryl-substituted oxa...

Scheme 24: Mechanisms of the formation of 1,2-diketones DK and of acetyl-substituted oxazole derivatives OX.

Scheme 25: Hydrogenolyses of benzyloxy-substituted β-ketoenamides KE52 and KE54 to 1,2-diketone DK14 and to di...

Scheme 26: Conversions of 2,4-dicyclopropyl-substituted oxazole OX7 into oxazole derivatives OX18–20 (PPA = po...

Scheme 27: Syntheses of vinyl and ethynyl-substituted oxazole derivatives OX21 and OX23 and their palladium-ca...

Scheme 28: Synthesis of C3-symmetric oxazole derivative OX28 and the STM current image of its 1-phenyloctane s...

Scheme 29: Condensation of 1,2-diketones DK with o-phenylenediamine to quinoxalines QU1–7 (CAN = cerium ammoni...

Scheme 30: The LANCA three-component reaction leading to β-ketoenamides KE and the structure of functionalized...
Tandem copper and photoredox catalysis in photocatalytic alkene difunctionalization reactions
- Nicholas L. Reed,
- Madeline I. Herman,
- Vladimir P. Miltchev and
- Tehshik P. Yoon
Beilstein J. Org. Chem. 2019, 15, 351–356, doi:10.3762/bjoc.15.30
- ligand sets might be required, as in enantioselective catalytic oxidation reactions or certain cross-coupling applications. We wondered, therefore, if catalytic loadings of copper(II) salts might be used in these reactions by adding a secondary terminal oxidant to turn over the intermediate copper(I
- dioxygen for this purpose is frequently exploited to effect synthetically useful copper-catalyzed aerobic oxidation reactions [27][28]. However, the use of molecular oxygen as a terminal oxidant presents unique challenges in photoredox chemistry. Triplet dioxygen rapidly quenches the excited state of most
- of Cu(TFA)2 to 10 mol % and assessed the effect of a series of alternate oxidants that have been used in other copper-catalyzed oxidation reactions. Most failed to produce significant quantities of the desired product (Table 1, entries 1–3). As expected, the use of oxygen as a terminal oxidant

Graphical Abstract

Figure 1: a) Photocatalytic oxyamination, b) photocatalytic diamination, and c) proposed mechanism for photoc...

Figure 2: Scope studies for dual-catalytic alkene difunctionalization using 2.5 mol % 3, 30 mol % Cu(TFA)2, a...






